Researchers from the University of Texas at Austin (UTA) may have solved the issue of dendritic growth in sodium-sulfur batteries— paving the way to a commercially viable alternative to lithium-ion.
Teams from the US institute have developed two methods of solving the dendrite problems that plague the technology.
One team has created an electrolyte that prevents the sulfur from dissolving, and enables a longer life cycle for the battery, with the scientists’ results showing a stable performance over 300 cycles.
The researchers found the electrolyte— designed by diluting a concentrated salt solution with an inert, non-participating solvent that preserves the “half-dissolved” state— prevented such the unwanted reactions at the electrodes.
Findings from team one were published in the Journal of the American Chemical Society.
A sodium-sulfur battery is appealing due to the relative cheapness and abundance of the materials.
Arumugam Manthiram, director of UTA’s Materials Institute and professor in the Walker Department of Mechanical Engineering, said: “I call it a dream technology because sodium and sulfur are abundant, environmentally benign, and the lowest cost you think of.
“With expanded electrification and increased need for renewable energy storage going forward, cost and affordability will be the single dominant factor.”
Previously, the intermediate compounds formed from sulfur would dissolve in the liquid electrolyte of sodium-sulfur batteries, and migrate between the two electrodes within the battery.
This shuttling has led to material loss, degradation of components, and dendrite formation.
Sodium anode research
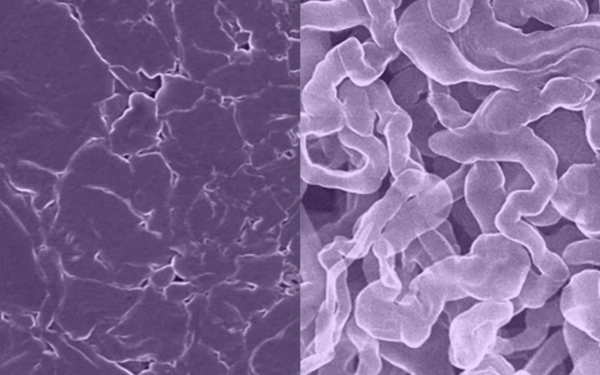
In separate research, a team led by David Mitlin developed an anode material called sodium antimony telluride intermetallic— Na metal composite (NST-Na).
Mitlin, who designed the new material, is a professor in the university’s Cockrell School of Engineering’s Walker Department of Mechanical Engineering and Applied Research Laboratory.
The material was made by rolling a thin sheet of sodium metal onto an antimony telluride powder, folding it over on itself, and repeating many times.
The process results in a uniform distribution of sodium atoms that makes it less likely to form dendrites or surface corrosion observed with existing sodium metal anodes.
This makes the battery more stable, allows charging comparable to a lithium-ion battery’s charge rate, and has a higher energy capacity than existing sodium-ion batteries, reported the team.
The study’s two lead authors Yixian Wang and Hui Dong— current and former graduate students in Mitlin’s laboratory respectively— fabricated the material.
The team published its results in the journal Advanced Materials.
Graeme Henkelman, a professor in UTA Department of Chemistry and its Oden Institute for Computational Engineering and Sciences, used a computer model to reveal what happens when individual sodium atoms interact with the new composite material NST-Na.
Henkelman said: “In our calculations, this composite binds sodium a little more strongly than sodium binds itself, which is the ideal case for having the sodium atoms come down and evenly spread out on the surface and prevent these instabilities from forming.
“This material is also exciting because the sodium metal anode theoretically has the highest energy density of any sodium anode.”
Mitlin, Wang and Dong have applied for a patent, along with UTA, on the new sodium metal anode material’s fabrication, structure and functionality.
Image: Scientists at the University of Texas at Austin have developed a new sodium metal anode for rechargeable batteries (left) that resists the formation of dendrites, a common problem with standard sodium metal anodes (right) that can lead to shorting and fires. Images were taken with a scanning electron microscope. Image credit: Yixian Wang/University of Texas at Austin.